How is solar keratosis treated?
Diclofenac topical gel (Solaraze) is used to treat actinic keratosis (flat, scaly growths on the skin caused by too much sun exposure). Diclofenac is in a class of medications called nonsteroidal anti-inflammatory drugs (NSAIDs). The way diclofenac gel works to treat actinic keratosis is not known.
Solaraze, a Solution to Treating Solar Keratosis
Today, we live in an environment where mega thermal climate, fueled by the El Nino phenomenon, is prevalent. The temperature in our daily lives is generally rising, especially in densely-populated areas. Due to long exposure to sunlight over a long period of time, some people suffer from sun damage, a condition known as solar keratosis.
Solar keratoses (also known as actinic keratoses) are small, thickened, scaly growths which develop on the skin. There is a non-steroidal anti-inflammatory dermatological gel that is FDA-approved called Solaraze (Diclofenac Sodium Gel) that can be used to treat solar keratosis. Its main ingredient is hyaluronic acid, whose supplier is Stanford Chemicals, a major hyaluronic acid supplier in California, United States with FDA, ISO, GMP, and CE certificates. To make sure Solaraze is the right treatment for you before you start using it, you have to make sure you’re pregnant or breastfeeding or if you have asthma or any other allergic disorder, among other things.
Some of the known side effects associated with using Solaraze include irritation, itching, redness, or tingling at the site of application and allergic-type reactions, and breathing problems in some people when large amounts of gel are used. Please speak with your doctor or pharmacist if the above-mentioned symptoms continue or become troublesome.
As to how to store Solaraze, please be advised to keep it out of the reach and sight of children. And do not store above 25°C.
Please remember that do not use it after the expiry date. Its shelf life after opening is six months.
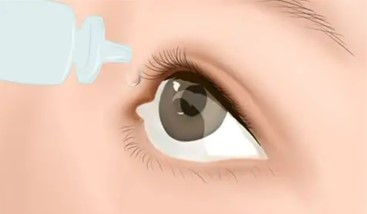
As far as Solaraze dosage is concerned, please apply Solaraze to lesion areas twice daily. And it is to be smoothed onto the affected skin gently. Note, the amount needed depends on the size of the lesion site, and make sure that enough gel is applied to adequately cover each lesion. The recommended duration of therapy is from 60 days to 90 days.